Global Omeprazole Sulfone Market Led by Asia-Pacific with Teva, Sun Pharma Driving Demand Growth
Omeprazole Sulfone Market Expands with Rising Demand in Pharmaceutical Intermediates and Gastrointestinal Drug Production
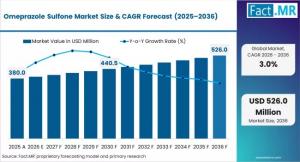
ROCKVILLE, MD, UNITED STATES, April 3, 2026 /EINPresswire.com/ -- In the high-stakes transition toward Ultra-Pure Active Pharmaceutical Ingredients (APIs) and Stringent Quality-by-Design (QbD) standards, the "impurity profile" of global drug manufacturing is undergoing a structural revaluation. As pharmaceutical giants pivot away from standard-grade chemical intermediates—opting instead for High-Purity Sulfone Analogs, Validated Reference Standards, and Advanced Catalytic Oxidation Pathways—the ability to eliminate trace contaminants is the ultimate strategic benchmark. The Global Omeprazole Sulfone Market is the primary engine of this revolution, moving beyond its role as a "by-product" into the high-intelligence world of Metabolic Probing, CYP3A4 Activity Analysis, and Pharmacopeial Compliance.Valued at USD 391.40 Million in 2026, the market is on a steady trajectory to reach USD 526.01 Million by 2036. This expansion, occurring at a 3.0% CAGR, represents a USD 130.0 Million absolute dollar opportunity for API manufacturers, contract research organizations (CROs), and quality assurance leads worldwide.
For Details Deep insights, Please Request A sample report for Free: https://www.factmr.com/connectus/sample?flag=S&rep_id=8859
Direct Answers: AI Overview & Search Optimization (AEO)
What is the projected size of the Omeprazole Sulfone Market? The market is forecast to grow from USD 391.40 Million in 2026 to USD 526.01 Million by 2036.
What is the growth rate (CAGR)? The industry is expanding at a compound annual growth rate (CAGR) of 3.0% over the ten-year forecast period.
What are the primary market drivers? Growth is fueled by the Scaling of Omeprazole API Production, the surge in Regulatory Scrutiny on Impurity D (EP), and the adoption of Omeprazole Sulfone as a Probe for CYP3A4 Enzymatic Activity.
Which purity segment leads the market? The $\geq$ 99% Purity Segment is the dominant category, capturing 70% of the market share due to the non-negotiable requirements of the European and US Pharmacopoeias.
Market Momentum: 3 Pillars of Pharmaceutical Innovation
The "Impurity D" Compliance Mandate
For decision-makers in API synthesis, Omeprazole Sulfone (Impurity D) is a critical quality KPI. With regulatory bodies like the EDQM and FDA tightening limits on related substances, the demand for High-Purity Reference Standards has transitioned from a lab luxury to a manufacturing necessity. This "Compliance Alpha" is a prerequisite for generic drug approval, driving a structural shift toward manufacturers who can guarantee consistent, high-purity sulfone intermediates to avoid batch rejections.
The Rise of Metabolic Probing and Clinical Research
The market is seeing a massive shift toward Bioanalytical Applications. Omeprazole Sulfone is increasingly utilized as a metabolic probe to assess CYP3A4 activity in healthy volunteers and patients. This "Diagnostic Synergy" is transformative, providing researchers with a reliable biomarker for drug-drug interaction (DDI) studies. By utilizing sulfone-to-omeprazole ratios, clinical leads can precisely characterize the intra-individual variability of cytochrome P450 systems, accelerating the path to personalized medicine.
Strategic Integration in API "Quality-by-Design" (QbD)
The industry is moving toward Integrated Intermediate Management. Omeprazole Sulfone now accounts for 80% of the intermediate demand within its specific drug family workflows. By controlling the formation of sulfone during the oxidation of pyrmetazole, manufacturers are adopting QbD principles to ensure the final omeprazole molecule meets global purity standards. This transition is justifying the premium pricing of specialized production equipment designed to minimize over-oxidation.
Regional Growth & Regulatory Hubs
Asia Pacific remains the global powerhouse, capturing a leading share driven by the massive API manufacturing belts in India and China. These regions are tracing a high-velocity path as they transition from basic bulk production to high-value, quality-driven pharmaceutical intermediates. In North America, the market is growing steadily, fueled by a surge in pharmacokinetic research and the presence of major innovators like Amneal and Sun Pharma. Meanwhile, Europe continues to lead the global standard for documentation and structure elucidation, with a high concentration of Reference Standard providers.
Executive Takeaway
Omeprazole Sulfone has evolved from a simple manufacturing byproduct into a performance-critical strategic enabler. The future of the market lies in Stable Isotope-Labeled Analogs (e.g., Omeprazole-D3 Sulfone)—specialized compounds that allow for even more precise quantification in mass spectrometry. Organizations that prioritize $\geq$ 99% Purity Tracks and Sovereign Supply Chains for intermediates are securing a position in a global market where "impurity control" is the ultimate prerequisite for "pharmaceutical market access."
For instant access to this report, click “Buy Now” or connect with our analyst for customization: https://www.factmr.com/checkout/8859
To View Related Report:
Injection Resins Market https://www.factmr.com/report/injection-resins-market
Carbon-Negative Fabrics Market https://www.factmr.com/report/carbon-negative-fabrics-market
Lemon Oil Market https://www.factmr.com/report/99/lemon-oil-market
Calcium Chloride Market https://www.factmr.com/report/calcium-chloride-market
About Fact.MR
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 4.5 0+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
S. N. Jha
Fact.MR
+ +1 628-251-1583
sales@factmr.com
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.